
Chronic wasting disease (CWD) is a fatal infectious disease that affects members of the Cervidae family, such as deer, elk, and moose. CWD develops when changes in the prion protein (PrP) cause a degeneration of the brain, giving it a spongy appearance. Other tissue that contains PrP protein will also be affected. Deer infected with CWD suffer from weight loss, excess salivation, loss of coordination, reduced fear response to humans, and slow death. This contagious disease was first discovered in mule deer in Colorado in the 1960s and has since spread across much of North America, reaching Illinois white-tailed deer in 2002 (Rivera et al., 2019).
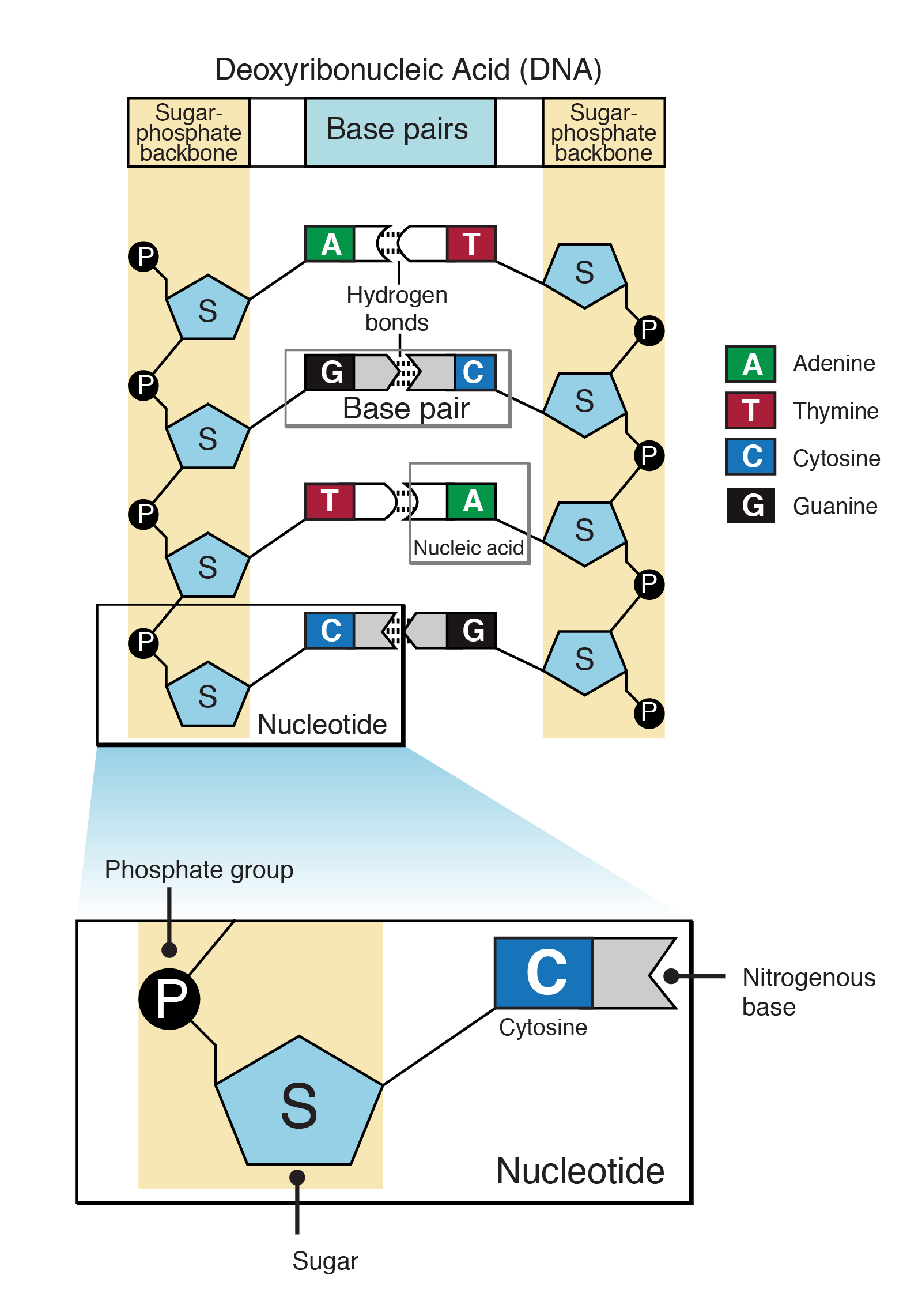
Like all proteins, PrP is encoded by a gene (stretch of DNA) consisting of DNA base-pair sequences (Figures 1 and 2). Over several decades, scientists have examined the prion protein gene and found that some deer species exhibit differences in the DNA sequence of the prion gene, which may reduce CWD susceptibility. In a 2018 study by Brandt and colleagues, the DNA sequence of the prion gene was analyzed in 2,433 white-tailed deer (Odocoileus virginianus), including 251 deer that tested positive for CWD (Brandt et al., 2018). Seven different DNA sequences were commonly found across the deer and the DNA sequences of the prion gene in CWD-positive and negative deer were compared. Two of the DNA sequences were found to be less common in CWD-positive deer (Brandt et al., 2018).

The PrP proteins generated from the different DNA sequences were also compared between CWD positive and negative deer. The five DNA sequences that were relatively common in CWD-positive deer all coded for the same type of PrP protein. By contrast, the two DNA sequences that were less common in the CWD-positive deer, each coded for a different PrP protein than the other five. It appears that the less common PrP proteins may have a protective effect on the deer, making them less susceptible to CWD (Ishida et al., 2020). Brandt and colleagues analyzed the geographic patterns of the two types of protective DNA sequences. They found that some regions of Illinois had somewhat higher proportions of genetically protected deer. This genetic advantage does not ensure that a deer is entirely resistant to CWD. Still, the two types of protective DNA sequences could elicit a high level of protection, suggesting that specific areas may see a lower prevalence of CWD in the population due to this genetic advantage. Furthermore, the frequency of this advantageous genetic variation may shape how CWD spreads throughout Illinois. While this speculation is not yet proven, further research and long-term monitoring of the genetics of wild deer across Illinois could give us more insight.

CWD is a terrible disease. There is no treatment or cure available, and it is 100 percent fatal. However, studies such as Brandt et al. (2018) can help managers assess risk and allocate limited resources in areas where the susceptibility to CWD is apparently higher. The discovery that genetics could impact animal susceptibility to CWD may allow us to predict how CWD could spread across areas or specific populations (Figure 3; Perrin-Stowe et al., 2020). Brandt et al. (2018) also show how genes could play an essential role in overall CWD herd immunity. These findings can certainly be a way forward to inform the management of CWD. We hope that this article and scientific paper will allow us to realize the power of research. For more information, please refer to the links below where you can access the scientific papers.
References:
- Brandt, A.L., Green, M.L., Ishida, Y., Roca, A.L., Novakofski, J. and Mateus-Pinilla, N.E., 2018. Influence of the geographic distribution of prion protein gene sequence variation on patterns of chronic wasting disease spread in white-tailed deer (Odocoileus virginianus). Prion,12(3-4), pp.204-215. https://www.tandfonline.com/doi/pdf/10.1080/19336896.2018.1474671
- Ishida Y., Tian T., Brandt A.L., Kelly, A.C., Shelton P., Roca, A.L., Novakofski, J.E. and Mateus-Pinilla, N.E., 2020. Association of chronic wasting disease susceptibility with prion protein variation in white-tailed deer (Odocoileus virginianus). Prion 14 (1), 214-225 https://doi.org/10.1080/19336896.2020.1805288
- Rivera, N.A., Brandt, A.L., Novakofski, J.E. and Mateus-Pinilla, N.E., 2019. Chronic wasting disease in cervids: Prevalence, impact and management strategies. Veterinary Medicine: Research and Reports, 10, p.123.https://www.dovepress.com/chronic-wasting-disease-in-cervids-prevalence-impact-and-management-st-peer-reviewed-fulltext-article-VMRR
- Perrin-Stowe, T.I., Ishida, Y., Terrill, E.E., Hamlin, B.C., Penfold, L., Cusack, L.M., Novakofski, J., Mateus-Pinilla, N.E. and Roca, A.L., 2020. Prion protein gene (PRNP) sequences suggest differing vulnerability to chronic wasting disease for Florida Key deer (Odocoileus virginianus clavium) and Columbian white-tailed deer (O. v. leucurus). Journal of Heredity, esaa040, https://doi.org/10.1093/jhered/esaa040
- Figure 1: Nucleotide definition. Courtesy: National Human Genome Research Institute. https://www.genome.gov/genetics-glossary/Nucleotide
- Figure 2: DNA sequencing definition. Courtesy: National Human Genome Research Institute. https://www.genome.gov/genetics-glossary/DNA-Sequencing
- Figure 3: By Averette – Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=4485643
Jacob E. Wessels earned his B.S. in Animal Sciences at the University of Illinois in 2018. He is currently a master’s student in the Novakofski & Mateus Chronic Wasting Disease Lab. His research involves studying a genetic resistance to Epizootic Hemorrhagic Disease in white-tailed deer. Upon graduation, he plans to pursue a career with the Illinois Department of Natural Resources as a wildlife biologist or conservation police officer.
Dr. Nelda Rivera's research focuses on the ecology and evolution of new and re-emerging infectious diseases and the epidemiology of infectious diseases, disease surveillance, and reservoir hosts’ determination. She is a member of the Wildlife Veterinary Epidemiology Laboratory and the Novakofski & Mateus Chronic Wasting Disease Collaborative Labs. She earned her M.S. at the University of Illinois at Urbana-Champaign and D.V.M at the University of Panamá, Republic of Panamá.
Dr. Adam Brandt was a Postdoctoral Research Associate with the Illinois Natural History Survey working in the Chronic Wasting Disease Collaborative Labs with Dr. Jan Novakofski and Dr. Nohra Mateus-Pinilla. He now is an Assistant Professor of Biology at St. Norbert College in DePere, Wisconsin. His research area is molecular ecology, specifically evolution, phylogenetics, population and conservation genetics.
Dr. Yasuko Ishida is involved in and studies chronic wasting disease in white-tailed deer in Illinois. Her main research focus is on conservation genetics and evolution as well as elucidating the association of genetics and diseases in animals. She earned her B.S., M.S. and PhD from the Kyushu University, Japan.
Dr. Alfred Roca is faculty in the Department of Animal Sciences of the University of Illinois at Urbana-Champaign. His research interests include comparative genomics of retroviruses and of mammals, including the prion protein gene in deer. Dr. Roca earned his Ph.D. at Harvard University, followed by postdoctoral studies at the Laboratory of Genomic Diversity in Frederick, Maryland.
Dr. Nohra Mateus-Pinilla is a veterinary Epidemiologist working in wildlife diseases, conservation, and zoonoses. She studies Chronic Wasting Disease (CWD) transmission and control strategies to protect the free-ranging deer herd’s health. Dr. Mateus works at the Illinois Natural History Survey- University of Illinois. She earned her M.S. and Ph.D. from the University of Illinois Urbana-Champaign.
Dr. Jan Novakofski studies prion diseases or infectious agents composed entirely of protein in animals such as “mad cow disease” and scrapie. His efforts are contributing to better understanding the genetics and transmission of these types of diseases to protect the health of animals and humans. He earned his B.S., M.S. and PhD from the University of Wisconsin, Madison.















Submit a question for the author